MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.

MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.
MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.

MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.

MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.
MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.

MEDICAL DEVICE & DRUG DELIVERY
Human Factors & Design Research Experts
We partner with medical device and pharmaceutical companies to design safer, smarter, and more effective products through rigorous human factors engineering and design research.
Our Experience
325+
Studies Completed
25+
Countries
8500+
Participants Evaluated
115+
Clients Served












Our Expertise
-

Design Research
We translate real-world use and unmet needs into clear direction for product design.
-
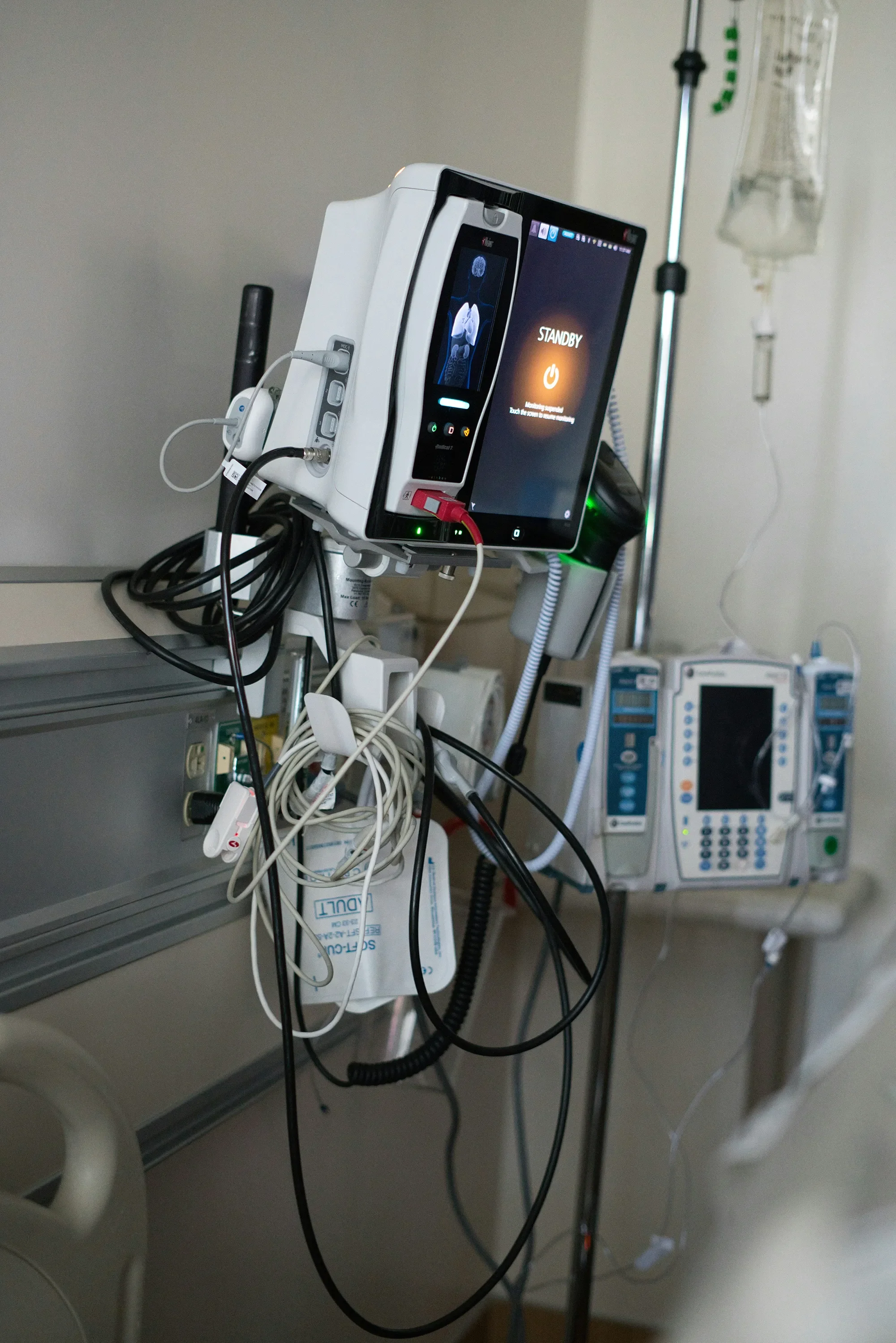
Usability Analysis
We analyze and document users, environments, interfaces, tasks, and training to inform and de-risk early design and support compliance.
-

Human Factors Documentation
We develop rigorous documentation needed to align with international standards and regulatory guidance.
-
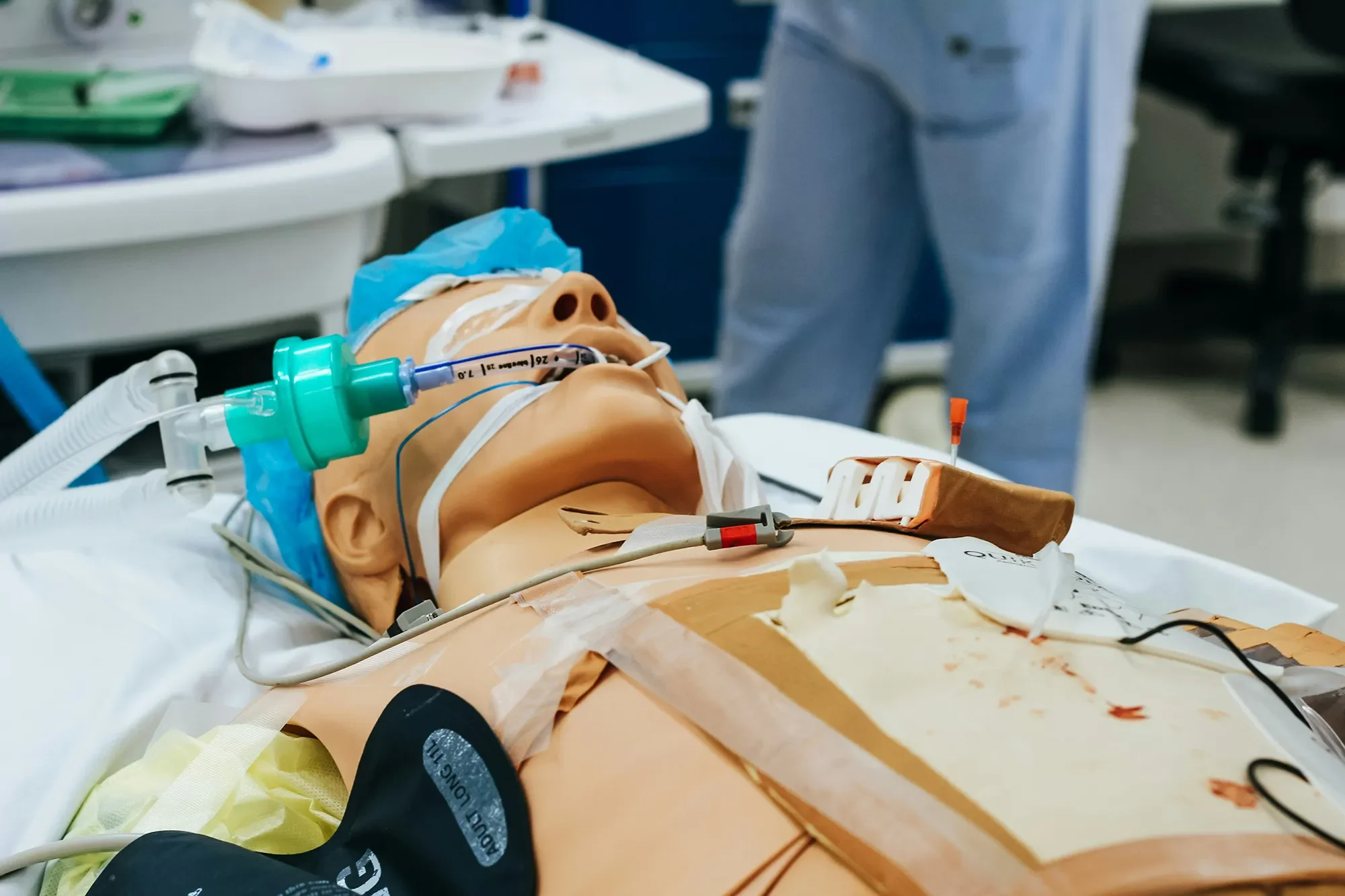
Formative Evaluations
We evaluate product use, identify usability issues, and highlight opportunities to mitigate risk and improve design.
-
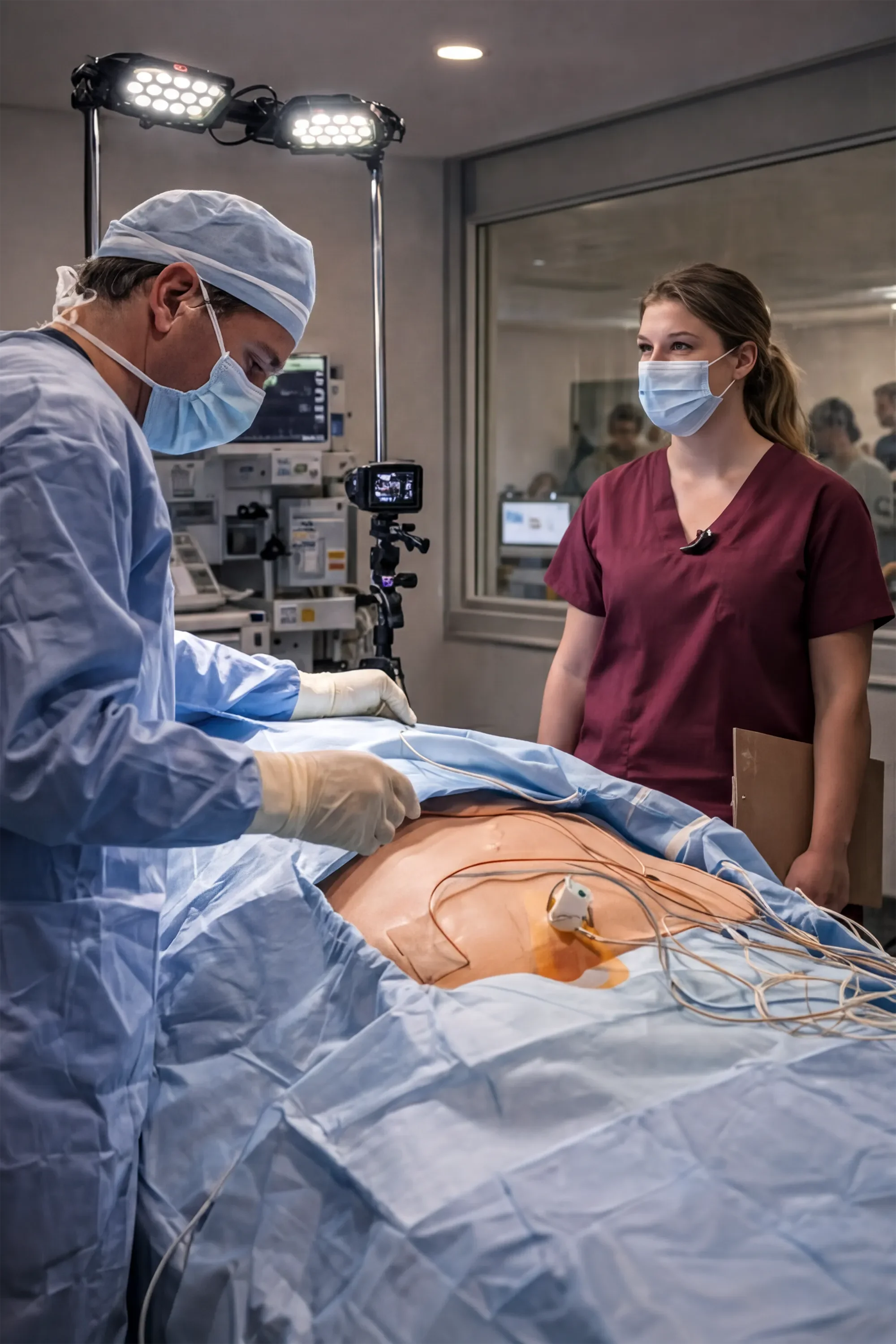
Validation Testing
We execute HF validation (summative) studies and generate clear reports and documentation for global regulatory authorities.
-
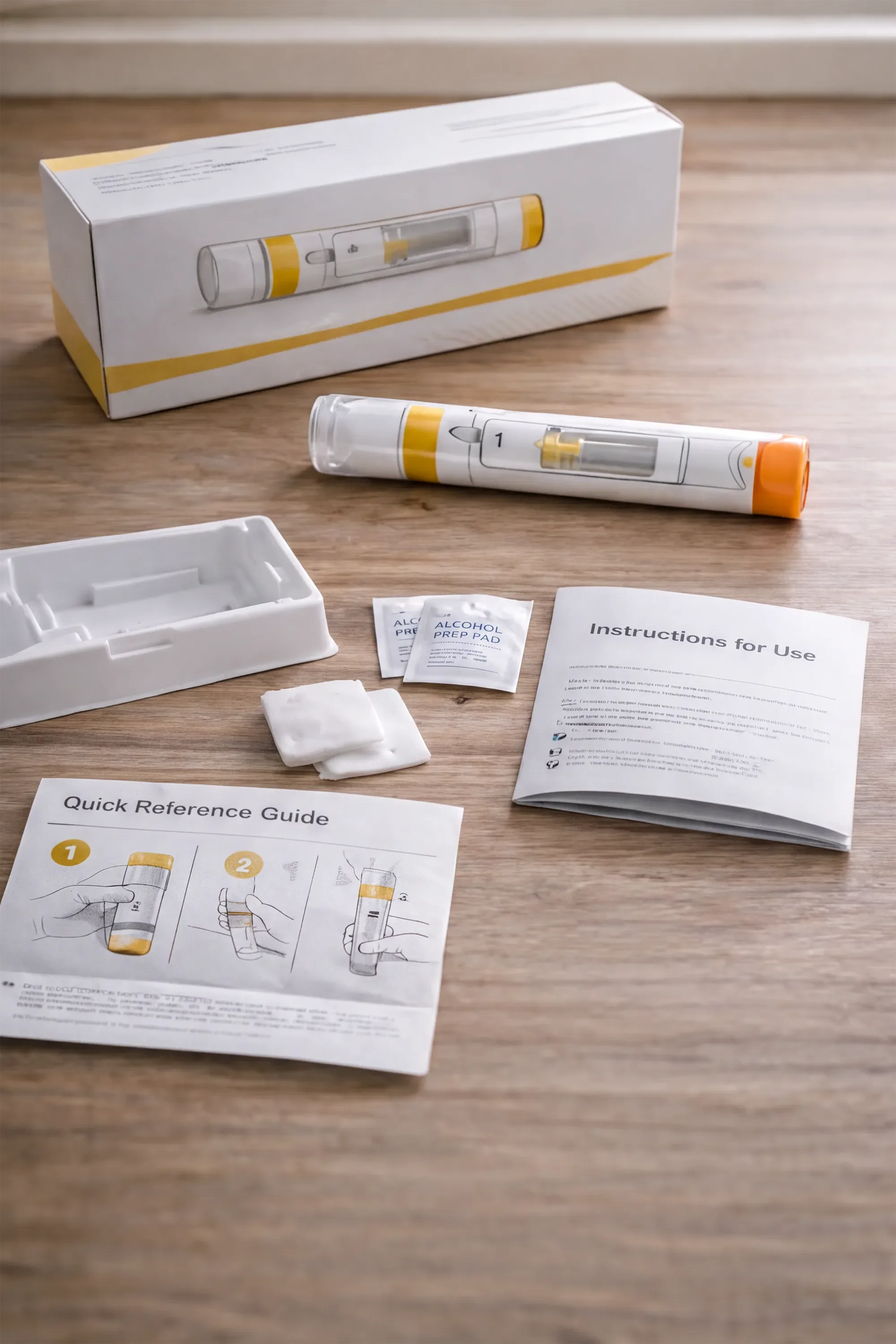
Instructional Design
We design labeling and instructional materials that effectively guide users and mitigate risk.
Clinical Specialties
Cardiovascular
Chronic & Home Care
ENT
Gastroenterology
General Surgery
Neurology
Oncology
Ophthalmology
Orthopedics & Spine
Pulmonology
Renal & Nephrology
Urology
Technology Areas
Ablation & Energy
Diagnostic & Imaging
Dialysis
Digital Health & SaMD
Drug Delivery
Endoscopy & Visualization
Implantable Devices
Life Support & Critical Care
Perfusion
Robotic Surgery
Surgical & Interventional
Wound Care
ABOUT US
Human factors and design research for medtech and pharma.
From early-stage innovators to global leaders, we help teams understand how healthcare products are used in the real world. Our senior team brings decades of experience across medical devices, drug delivery systems, diagnostics, and digital health, combining design research with risk-based human factors engineering to guide development. We deliver clear, actionable insights that inform design, reduce risk, and enable safer, more effective products.
How We Work
End-to-end or targeted support.
We can manage your project end-to-end or simply plug-in where needed.
Study design and planning
Recruitment and honoraria*
Research facilities and logistics*
Study moderation and notetaking
Analysis, reporting and documentation
Insight synthesis and recommendations
*Via specialized & qualified partners
An extension of your team.
We provide leadership when you need it and support when you don’t.
In-depth product onboarding and knowledge transfer process
Act as both a collaborative partner and subject matter expert
Align with existing processes and templates – or bring our own
Clear communication and accountability
Flexible ways to partner together.
We offer a variety of engagement models to fit your needs.
Predictable fixed-fee projects
Hourly time and materials
Open-ended monthly support
Staff augmentation
Fractional HF leadership

Confident Decisions Start With Clear Insight
Whether you are refining a concept, validating a design, or preparing for regulatory submission, Pulse HF provides the human factors expertise and research depth needed to move forward with certainty.